SARS-CoV-2 & Flu A/B Test
a rapid microfluidic immunofluorescence assay designed to optimize patient and clinical flow. Used with the LumiraDx Platform, the LumiraDx SARS-CoV-2 & Flu A/B test provides actionable and lab-comparable results in 12 minutes for patients suspected of Flu and/or COVID-19.
Qualitative SARS-CoV-2 & Flu A/B test
The LumiraDx SARS-CoV-2 & Flu A/B test is an automated rapid microfluidic immunofluorescence assay for the qualitative detection and differentiation of SARS-CoV-2, Influenza A and/or Influenza B viral antigens directly from anterior nasal swab samples.
Near-patient testing
As an automated in vitro diagnostic test, it is made for near-patient testing with the LumiraDx Instrument.
Purpose
The test can be used as an aid in the differential diagnosis of SARS-CoV-2, Influenza A and Influenza B.
Benefits

Optimize patient and clinical flow
Verify potential infection quickly with 3 actionable results from one patient sample:
- Test Flu A, Flu B and COVID-19
- Sample type: nasal swab
- Prepared sample can also be used with LumiraDx SARS-CoV-2 & RSV test*
- SARS-CoV-2 Ag: - PPA** up to 95.5% - NPA* up to 99.0%
- Flu A: - PPA 83.3% - NPA 97.6%
- Flu B: - PPA 80% - NPA 94.9%
- Time to result: 12 minutes
- Storage at room temperature (2°C to 30°C)
*Swab must be validated for use with both tests - refer to swab technical bulletins
**Based on 0-12 days since symptom onset data. Refer to SARS-CoV-2 & Flu A/B Product Insert for full clinical data

Workflow
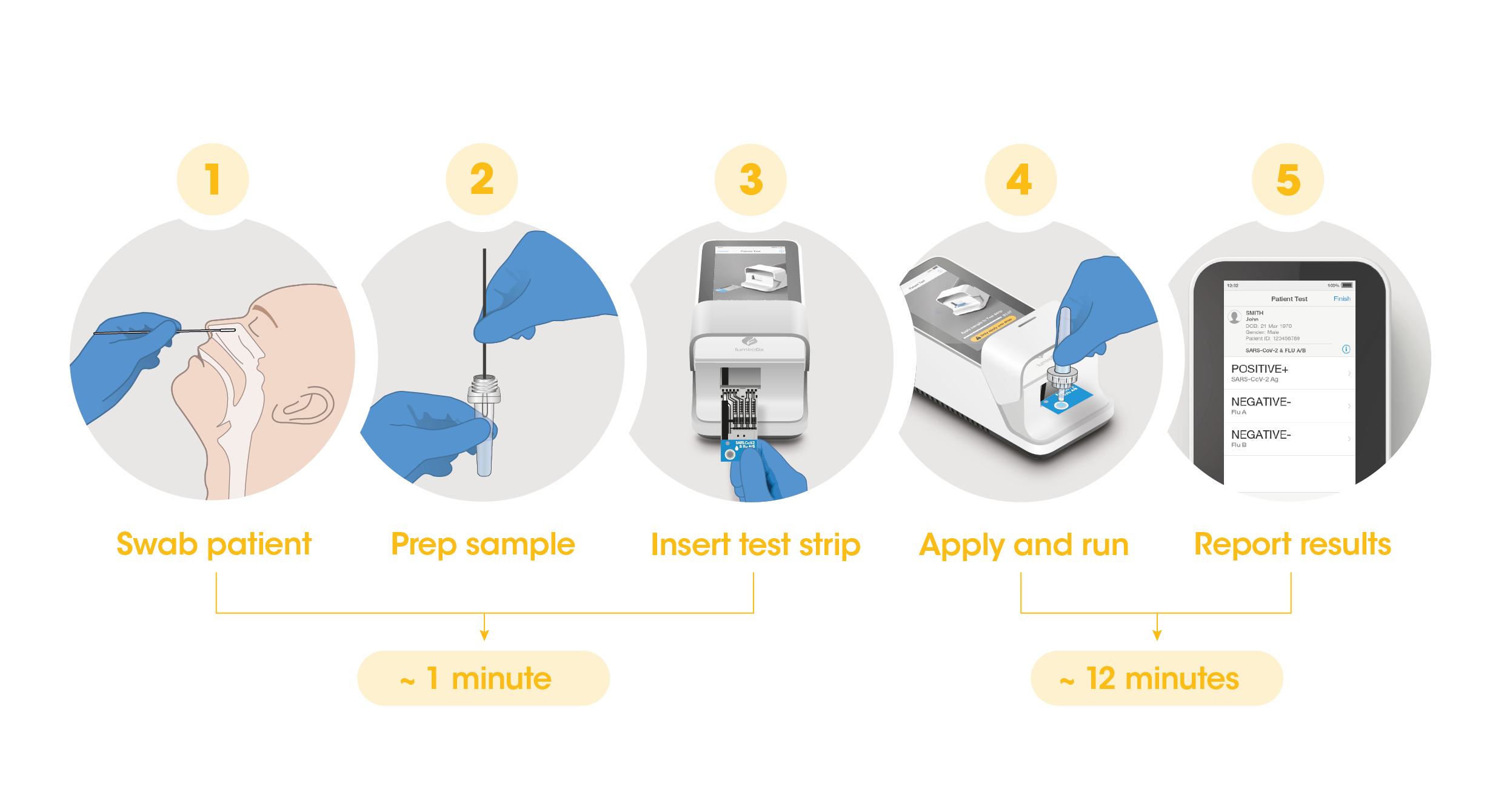
The workflow process is comprised of a simple sample prep along with step-by-step guidance of the Instrument to report a patient result in 12 minutes from sample application.
The Instrument and test strips are integrated with several quality control checks to ensure the Instrument and test are functioning correctly for every test run.
HOW TO USE VIDEOTest Performance
Clinical performance
In clinical studies, the LumiraDx SARS-CoV-2 & Flu A/B test demonstrated up to 95.5%, 83.3% and 80.0% positive agreement versus RT-PCR for detection of SARS-CoV-2, Flu A and Flu B respectively.
Analytical performance
The final limit of detection of the LumiraDx SARS-CoV-2 & Flu A/B test was determined to be the lowest concentration resulting in positive detection of at least 95% of replicates. Click above for the LoD values for different virus materials.
Cross reactivity
SARS-CoV-2 & Flu A/B was found not to cross-react with a panel of organisms and viruses including several human coronaviruses. See LumiraDx SARS-CoV-2 & Flu A/B Product Insert for full details.
Product Documentation
SARS-CoV-2 & Flu A/B Test:
SARS-CoV-2 & Flu A/B Test

