fast
lab-accurate
results where
you need them

Our test strips use next-generation actively controlled microfluidics to drive the onboard chemistry and to deliver lab-comparable results.
The Platform allows for multiple sample types, including direct fingerstick and swab, and only requires one drop to run a test.
A simple and intuitive user interface provides step-by-step guidance through the easy to learn test workflow.


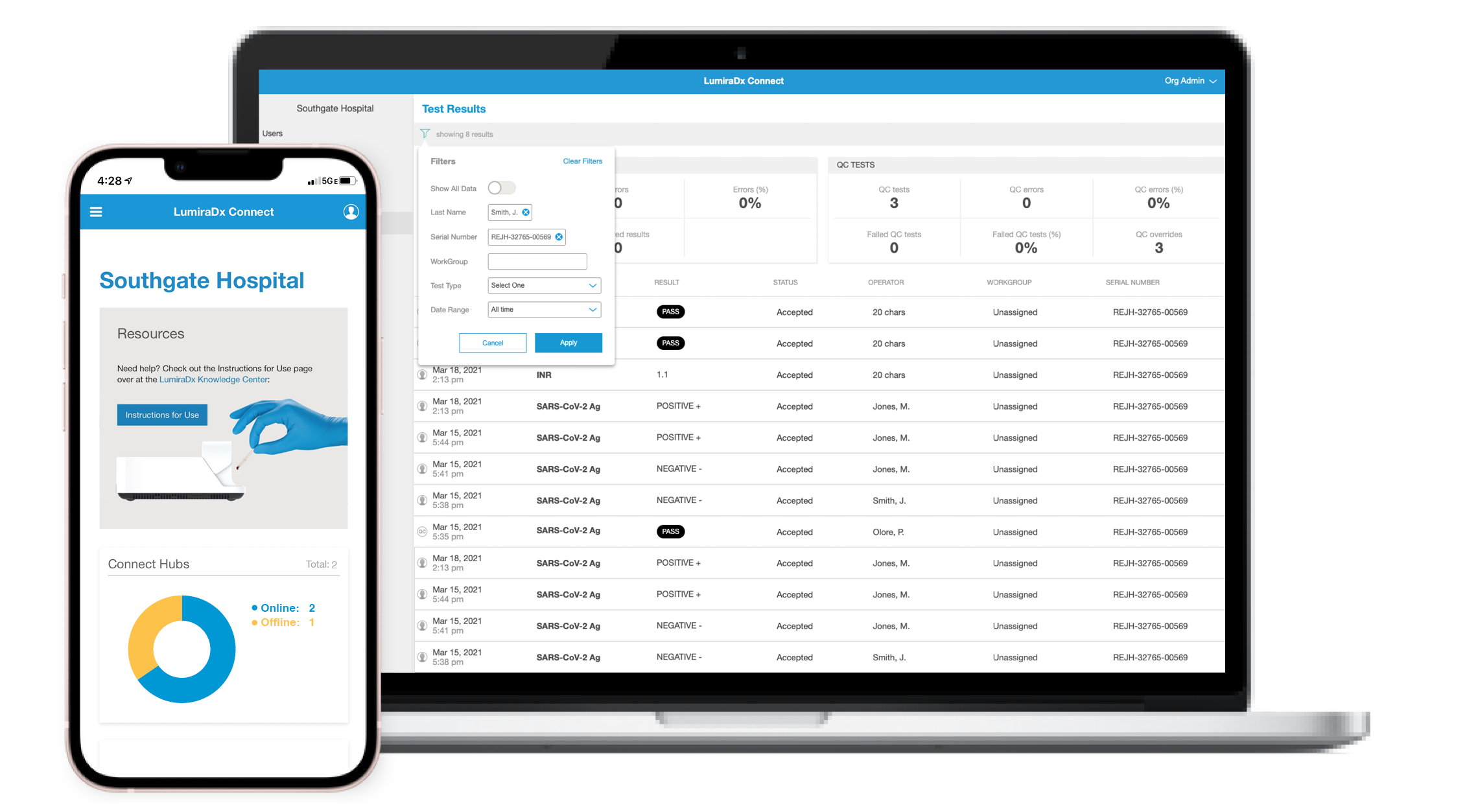
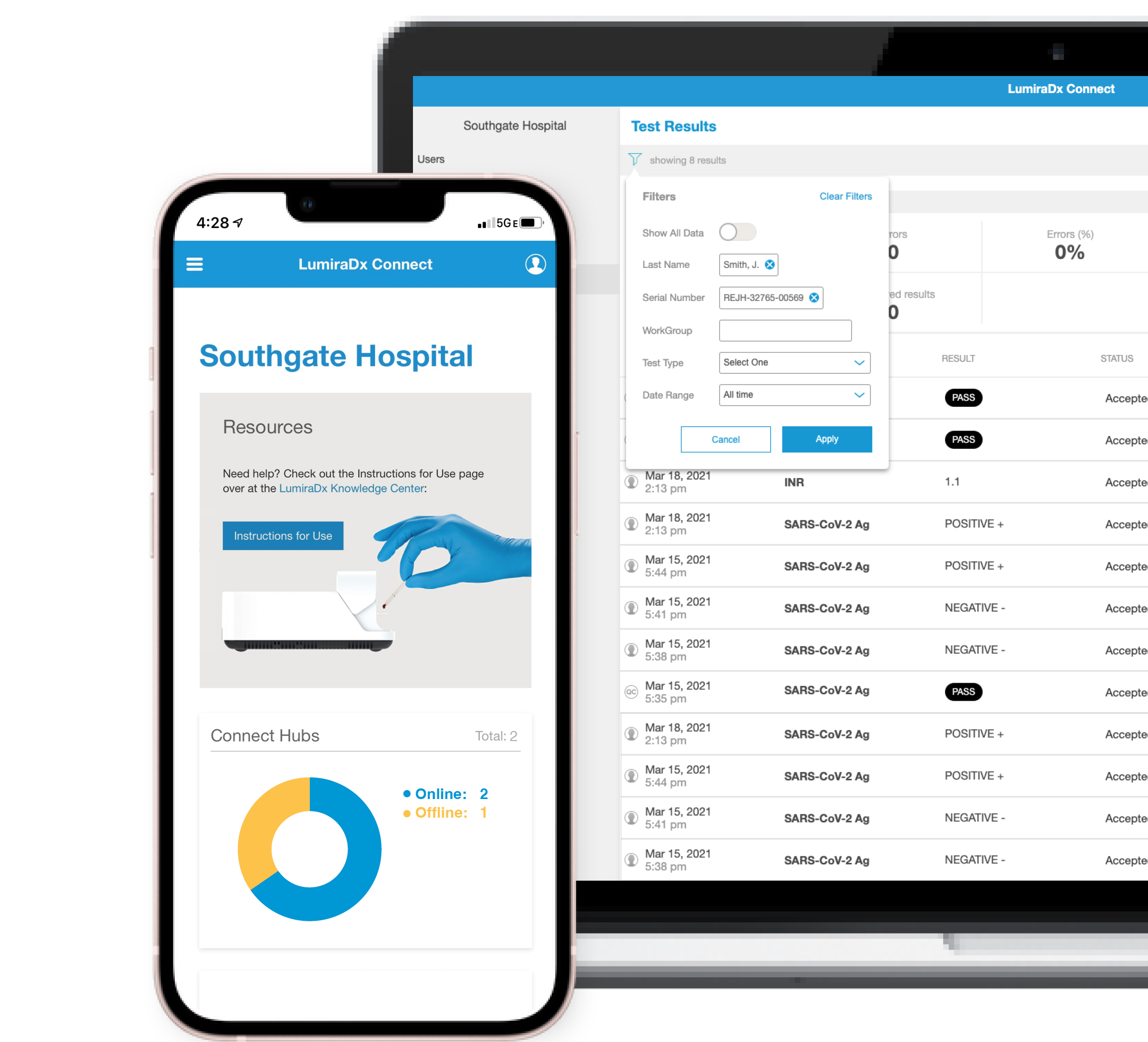
Connect to your devices
Connect Manager® is a cloud system with online reports that are easily accessible from any device. Manage and configure instruments, view results and simplify the provisioning of work groups remotely from any mobile, tablet or PC
Platform training
Get the most out of your point of care Platform with these training videos
Watch the videosConnectivity training
Learn to easily connect devices and get the full potential of the Connect Manager system
Watch the videosEnhance your workflow
Discover the LumiraDx products that can that help maximize your experience with your smart diagnostic testing solution
Instant Printer

Barcode Scanner

Connect Hub

Carry Case

Related publications
White PaperLink
Evaluation of LumiraDx RSV/ Influenza/ SARS-CoV-2 tests in pediatric emergency departmentsNewsLink
Roche closes acquisition of LumiraDx’s Point of Care technology to expand access to diagnostic testing in primary careWhite PaperPDF
St Malo Evaluation of LumiraDx Analyzer for decentralized CRP testing