Live Event
COVID-19 Testing
"It’s time to change how we think about the sensitivity of testing for COVID-19."1
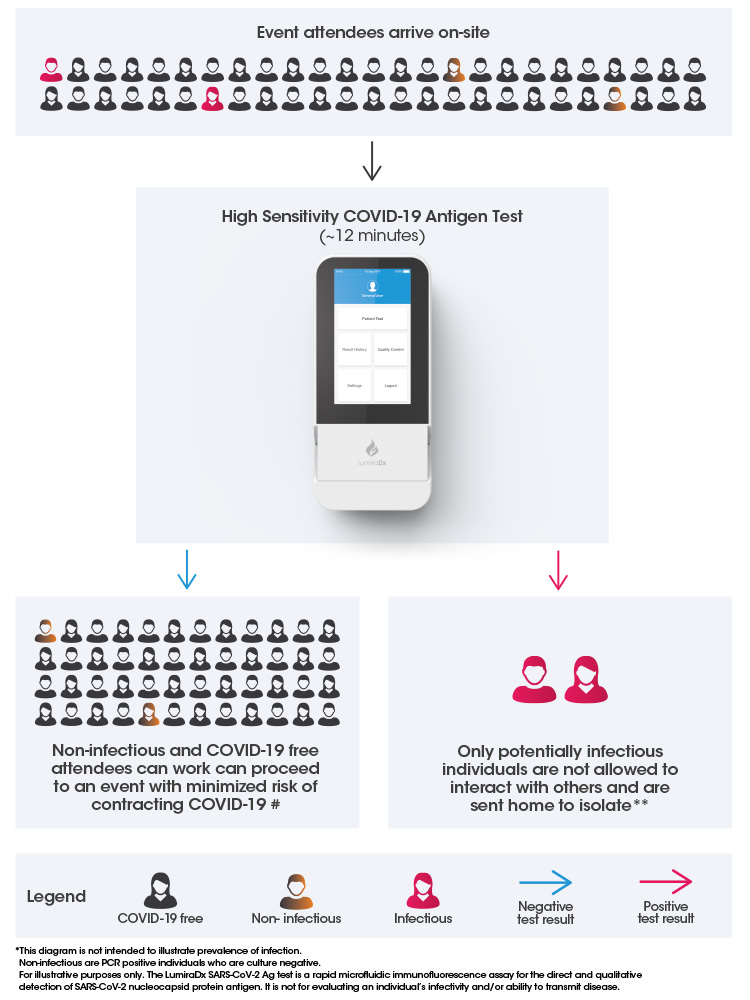
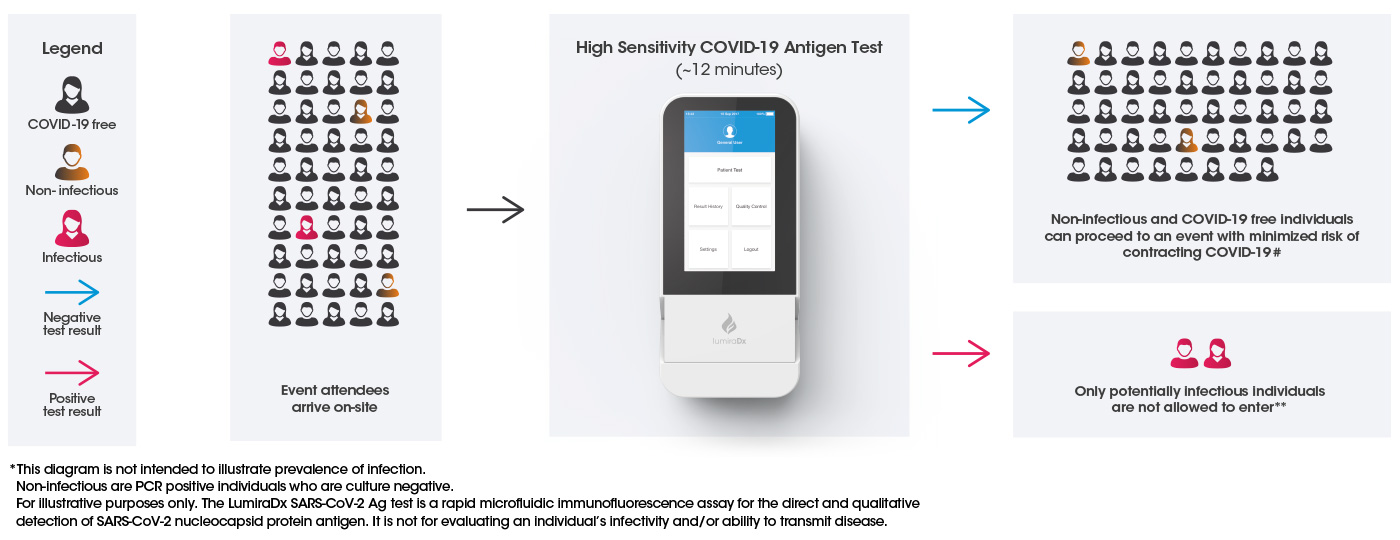
Accurate and rapid pre-event testing to safely reopen cultural, sports and leisure events
Pre-event testing is necessary to reduce risks of positive Covid-19 cases at events so that more activities can resume safely.
Pre-event testing for COVID-19 helps to reduce the risk of potentially infectious individuals transmitting the virus at an event, reducing the likelihood of an outbreak situation.
Uncompromised rapid testing with the LumiraDx Platform delivers fast, accurate antigen testing to give organizers the assurance they need that.
a) They can identify potentially contagious spreaders pre-event
b) Attendees can easily and quickly be tested in advance of the event
LumiraDx provides a range of testing solutions:
- Rapid and connected test results in minutes
- Portable and flexible solutions for on or off-site testing setup
- Affordable and scalable testing
COVID-19 testing
CTSB enables the next step of normal life to resume
with the LumiraDx SARS-CoV-2 Pool Test
Watch here
The benefits of uncompromised on-site testing with LumiraDx
- Less-sensitive lateral flow antigen tests2-4 increase the risk of an infectious attendee getting a negative result.
False negatives leave the door open for contagious persons to slip through (i.e., goes to an event rather than flagged for isolation) - ‘Swab and send’ delays time needed to get to actionable results
- PCR can still be positive long after period of infectivity3
#Negative results do not rule out SARS-CoV-2 infection and should be considered in the context of an individual’s recent exposures, history and presence of clinical signs and symptoms consistent with COVID-19. Results should not be used as the sole basis for treatment or case management decisions, including infection control decisions
**Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
SARS-CoV-2
Testing solutions
Easy to implement on-site at point of care
- Room Temperature Storage (2-30˚C)
- Results in 12 minutes
- Compact and portable instrument with connectivity options
Clinically sensitivity Assays
- Optimal sensitivity to rapidly detect potentially infectious individuals to prevent onward transmission
Adaptable and scalable solutions
- Individualized and scalable testing options to save time and costs
- One platform for both group and individual testing
SARS-CoV-2 Ag Test
- LumiraDx SARS-CoV-2 Ag test demonstrates high sensitivity at Ct<336 and can detect 10-30% cases potentially missed by antigen lateral flow tests7-11
- PCR can still be positive long after period of infectivity, leading to misidentification of infective individuals5. SARS-CoV-2 Antigen correlates with culture better than PCR for aiding in the identification of infective individuals12
- LumiraDx SARS-CoV-2 Ag Test provides high detection of potentially infective individuals
SARS-CoV-2 Pool Test
- Test up to 5 people using 1 test
- Maximise resources and scale up testing: Pooled testing can improve access to testing by significantly reducing resources (tests, operators, instruments) required on a per patient basis
- Save time and cost: Pooled testing can result in savings in both time and cost
LumiraDx Connect
- LumiraDx Connect is an intelligent, integrated connectivity platform that uses the latest cloud-controlled technology for use with LumiraDx Instrument.
- LumiraDx Connect ensures easy, secure transfer of results from LumiraDx Instrument to Connect Manager.
- Can be used with mobile phones, tablets or PCs
1: Michael J. Mina, etal., Rethinking Covid-19 Test Sensitivity — A Strategy for Containment, N Engl J Med 2020; 383:e120
2: Report of the „Bundesamt für Gesundheit“ Switzerland 2020 (available upon request)
3: Kohmer et al., 2021. J Clin Med Jan 17;10(2):328.
4: Krüger et al., 2021 (https://www.medrxiv.org/content/10.1101/2021.03.02.21252430v1)
5. www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html
6: Drain PK, Ampajwala M, Chappel C, et al. A Rapid, High-Sensitivity SARS-CoV-2 Nucleocapsid Immunoassay to Aid Diagnosis of Acute COVID-19 at the Point of Care: A Clinical Performance Study. Infect Dis Ther. 2021;10(2):753-761.
7: Linares et al, Panbio antigen rapid test is reliable to diagnose SARS-CoV-2 infection in the first 7 days after the onset of symptoms, J Clin Virol. 2020 Dec; 133: 104659
8: Gannon C.K. Mak, Stephen S.Y. Lau, Kitty K.Y. Wong, Nancy L.S. Chow, C.S. Lau, Edman T.K. Lam, Rickjason C.W. Chan, Dominic N.C. Tsang, Evaluation of rapid antigen detection kit from the WHO Emergency Use List for detecting SARS-CoV-2, Journal of Clinical Virology, Volume 134, 2021, 104712, ISSN 1386-6532, https://doi.org/10.1016/j.jcv.2020.104712.
9: Young S, Taylor SN, Cammarata CL, Varnado KG, Roger-Dalbert C, Montano A, Griego-Fullbright C, Burgard C, Fernandez C, Eckert K, Andrews JC, Ren H, Allen J, Ackerman R, Cooper CK. 2021. Clinical evaluation of BD Veritor SARS-CoV-2 point-of-care test performance compared to PCR-based testing and versus the Sofia 2 SARS antigen point-ofcare test. J Clin Microbiol 59:e02338-20. https:// doi.org/10.1128/JCM.02338-20
10: Osterman, A., Baldauf, HM., Eletreby, M. et al. Evaluation of two rapid antigen tests to detect SARS-CoV-2 in a hospital setting. Med Microbiol Immunol 210, 65–72 (2021). https://doi.org/10.1007/s00430-020-00698-8
11. Labcorp EUA – Histogram of Cts, https://www.fda.gov/media/136151/download
12: Andrew Pekosz et al., Antigen-Based Testing but Not Real-Time Polymerase Chain Reaction Correlates With Severe Acute Respiratory Syndrome Coronavirus 2 Viral Culture, Clinical Infectious Diseases, 2021;, ciaa1706, https://doi.org/10.1093/cid/ciaa1706
BetterHealthExperiencesOutcomes
Supporting healthier lives, for individuals, communities and wider society
Enabling responsive, personal relationships between patients and care teams.
Controlling and reducing costs to help ease pressure on healthcare budgets.